Saturated and Superheated Steam - Why use saturated steam?
Saturated steam, a condition in which steam contains no more energy than it took to evaporate water at a given pressure, is characterized by its temperature. To every generation pressure there is a corresponding saturation temperature-pressure relationship; saturation temperature for each generation pressure can be read from a steam table. If steam is heated above the saturation temperature, we speak of superheated steam. Whenever steam is cooled below its saturation temperature, it condensates releasing the latent energy it took to evaporate. When using steam to heat other media, it is thus desirable to deliver it to the contact surface at saturation temperature, so that condensation occurs inmediately and heat release is maximized. Superheated steam reaching a heating surface will first cool down, releasing only part of its energy; only when its temperature has come down to the saturation value, it will condensate. Thus the heating surface, i.e. a heat exchanger, will work only partially at full power.
In some cases, it is a requirement not to exceed a certain temperature when heating a medium, lest the product will be damaged. Delivering saturated steam at the adequate pressure guarantees that medium temperature will never exceed the corresponding saturation temperature.
Pressure reducing valves (PRV) cause superheat
Steam, as it is produced in a boiler, is naturally saturated. But generally, steam is not generated very close to the places where it will be consumed and so it will travel first considerable lengths trough pipes and ellbows, loosing pressure and energy on the way and condensating partially. Therefore, to minimaze losses, it has to be generated at a higher pressure than required by the end consumers. On reaching destination, pressure will typically have to be reduced to the values needed by end consumers; this is generally done by a pressure reducing valve (PRV), installed just before the consumer.
A PRV reduces pressure by solely expading steam volume, there is no work done and internal energy of steam remains practically constant (in reality some work is done at overcoming the electrostatic attraction of steam molecules), and steam leaves the PRV at almost the same temperature as it entered. We have then superheated steam, because pressure went down but not its temperature.
Steam conditioning valves (SCV)
An approach at delivering saturated steam at end the user's site is the steam conditioning valve; it consists of a pressure reducing valve and a water injection device. A pressure and a temperature sensor are installed downstream; a pressure PID controls the PRV, while a temperature PID controls the water injection. In practice, the temperature PID hast to be set at several degrees above saturation temperature to avoid water in the line downstream. On every start-up, the SCV needs a certain time to stabilize, in wich wet steam may be delivered first.
GEYSIR II
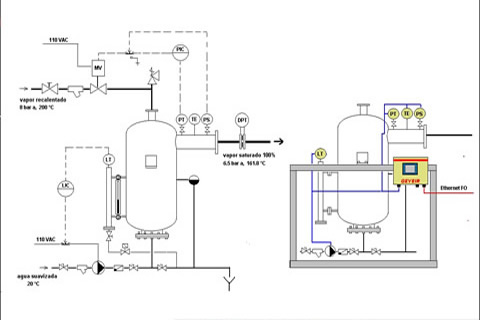
An effective approach is GEYSIR II , which, behaving as a low pressure boiler, delivers 100% saturated steam at consumer's site. The principle is the following: high pressure (possibly wet) steam is expanded to bring it down to the desired pressure; it then enters a pressure vessel in wich a difuser disperses the steam in thousands very fine streams through a cooling water bath, from which it emerges as completely saturated steam. Condensate is fed in to replaced the cooling water spent (converted into low pressure steam) in the process. GEYSIR II works thus with a pressure PID and a level PID. Advantages are:
- - it delivers 100% saturated steam from start-up, with no stabilization time
- - after a process shut-down, it keeps the vessel pressurized and delivers saturated steam inmediately again when it is requested.
- - it converts wet steam into saturated steam
- - it converts superheated steam into saturated steam
Downloads:
GEYSIR II - Introduction
GEYSIR II - Technical Data
GEYSIR II - Applications
GEYSIR II - Flyer